Discovering the Future of Cardiovascular Medicine
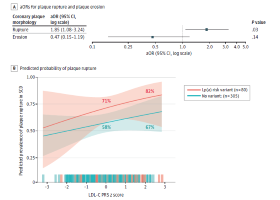
Pathological Analysis of Lipoprotein(a) Risk Variants and Culprit Coronary Thrombotic Lesions
CVPath’s research letter, led by Dr. Tatsuya Shiraki, was recently published in JAMA Cardiology. This paper examines the association between…
Dr. Virmani Attends Society for Cardiovascular Angiography & Interventions (SCAI) Meeting
Last week, Dr. Renu Virmani attended the Society for Cardiovascular Angiography & Interventions (SCAI) meeting in Montreal, Canada. She was…
CVPath Research Fellow Dr. Kazuhiro Fujiyoshi Presents at ACC 2026
Dr. Kazuhiro Fujiyoshi, a research fellow at CVPath, recently attended the 2026 American College of Cardiology (ACC) Annual Scientific Session…
Consultation Services
The CVPath Institute facility was designed to provide expert translational bench-to-bedside research services. Our team of experts has decades of experience in medical device evaluation and is able to provide guidance and advice on device safety, mechanism of action studies, or FDA responses. These services are provided all around the globe and for companies from start-ups to Fortune 100 manufacturers.

Comprehensive histology services from basic to advanced.

CAP-accredited and CLIA-approved laboratory to meet your diagnostic needs.

An internationally recognized research team offers study design consultation

A broad range of medical device experience to enhance your research needs

A full array of diagnostic services for autopsy and surgical specimens

Offering comprehensive microscopy technologies including SEM, OCT and Micro-CT.
Our Experts
The Institute’s research team consists of world class cardiologists, pathologists, scientists, and other highly skilled technical staff who have a deep understanding of cardiovascular pathology.

President and Chief Scientific Officer

President Emeritus and Founder

Associate Director

Chief of Anatomic Pathology

Staff Pathologist

Principal Research Scientist

MicroCT Project Manager

Staff Pathologist
Our Research Capabilities and Facilities

A state of the art lab purposely designed for cardiovascular studies

Ability to perform advanced molecular assays and experiments

A state of the art research laboratory located along the I-270 Technology Corridor

One of the largest tissue biobanks of its kind

Rapidly expanding genomic research capabilities

Extensive registry of cardiovascular specimens and correlating diseases
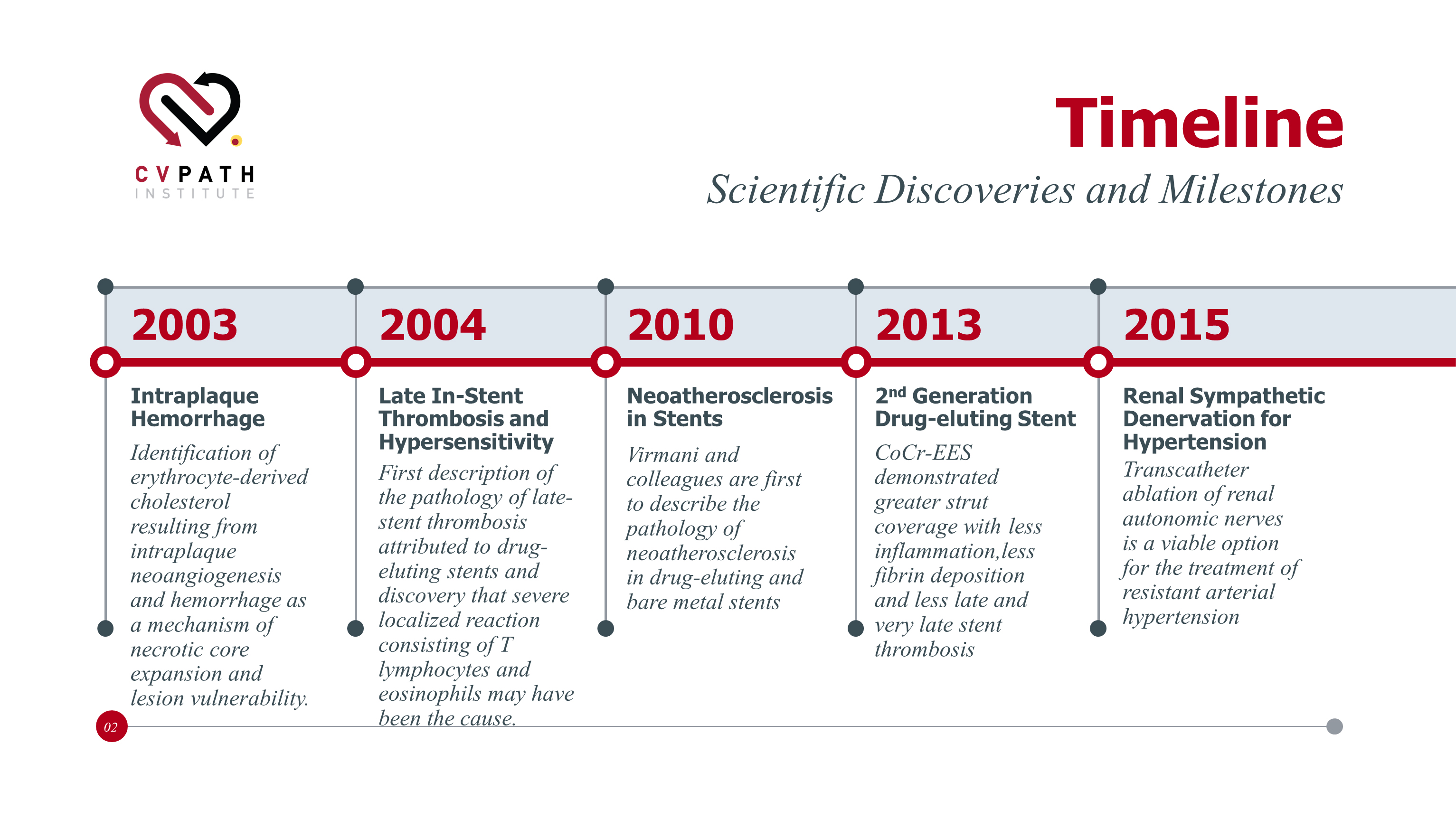
30 Years of Discoveries and Milestones
Pioneering discoveries in the cardiovascular field from the researchers of CVPath Institute.
Discoveries in the 1990’s
Discoveries in the early 2000’s
Recent discoveries

The latest from CVPath
Educational Services
CVPath Institute offers educational opportunities to train tomorrow's scientific leaders through workshops, fellowships and internships. The institute provides expert training in advanced pathology techniques to promising researchers in various stages of their educational and vocational careers.

Our “Hands on Hearts” workshop is offered at various cardiovascular conferences throughout the year.

A highly competitive fellowship program in the field of cardiovascular research and pathology.

Undergraduate opportunities to learn in a research environment.
Contact Us
Interested in learning more about how we can help your organization? Reach out to us and we'll be in touch with you shortly.